Inflammacheck®
Non-invasive point-of-care testing with results available two minutes later
For the first time, thanks to our simple to use and cutting-edge technology, physicians can pinpoint respiratory conditions through the measurement of hydrogen peroxide (H2O2) in exhaled breath condensate.
Monitor respiratory condition and inflammation through normal breathing
With Inflammacheck®, advanced factory calibrated sensors monitor respiratory condition and inflammation through normal, tidal breathing. With our light-weight and simple design, tests can be carried out quickly and easily on all patients, regardless of the severity of their disease.
With Inflammacheck®, you can measure and log five different biomarkers in just one breath:
- H2O2 contents
- CO2 contents
- Exhaled breath flow
- Exhaled breath temperature
- Exhaled breath relative humidity
Inflammacheck® adheres to the technical standards published by the ERS (European Respiratory Society)* and ATS (American Thoracic Society) in 2017, and thus defines an ERS/ATS technical standard compliant standardized methodology.
*Footnote: As recommended by the ERS Technical Standard for testing in EBC; Horváth, et al. ‘A European Respiratory Society technical standard: exhaled biomarkers in lung disease.’ Eur Respir J 2017; 49: 1600965, https://doi.org/10.1183/13993003.00965-2016


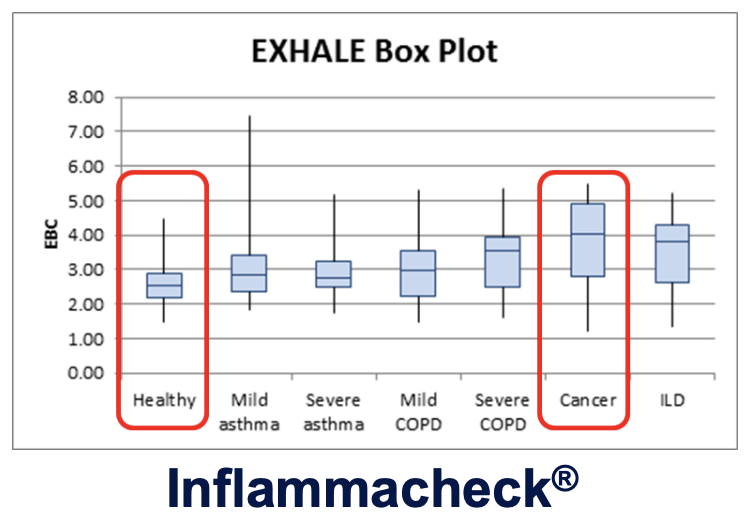
H2O2 is a relevant biomarker in a range of severe, respiratory diseases, including Lung Cancer
High levels of hydrogen peroxide (H2O2) have been consistently detected in the breath of individuals diagnosed with lung cancer at all stages. This discovery offers a potential non-invasive method for early detection and monitoring of the disease.
Clinical evidence indicates that testing for H2O2 with Inflammacheck®
can be applied in a wide range of severe, respiratory conditions – not the least Lung Cancer.
This discovery presents an exciting opportunity for cost-effective and time-efficient pre-screening, enabling the identification of individuals at high risk of developing lung cancer.
By enhancing detection rates and supporting early diagnosis, it holds great potential for improving overall outcomes in lung cancer management.
Proposed initial Use Case for Inflammacheck® in Lung Cancer screening
- Inflammacheck® may be applied at POC as a pre-screening test, in order to better qualify candidates for LDCT scanning for Lung Cancer
- Inflammacheck® will thus feed into the risk assessment conducted to identify those at highest risk of having Lung Cancer
- Applying Inflammacheck® is expected to:
- Help identifying LDCT scanning candidates most likely to have Lung Cancer, thereby increasing the detection rate and thus overall health economy
- Help identifying LDCT scanning candidates at early disease stages, not detected through other risk parameters, thereby enabling a higher detection rate at earlier, treatable disease stages

Use case: Addressing unmet need for Point of Care test to support qualification of candidates for Low-Dose CT scanning (LDCT) in Lung Cancer screening
- Pain Points
- High treatment and societal costs, high mortality
- 75% at advanced disease (stg. III/IV) at time of diagnosis
- 1-3% LDCT scan detection rate in high-risk individuals
- Bottlenecks in LDCT scanners and qualified radiologists

Our fully automatic Inflammacheck®, can be used without any specialist training
A tool to collect accurate, insightful information on lung condition and potential airway inflammation in only 5 minutes.
Non-invasive point-of-care testing means physicians can gather a full sample after 1-3 minutes, with results available two minutes later.
Inflammacheck® analyses H2O2 levels in exhaled breath to detect and track possible oxidative stress and lung inflammation.
Our ground-breaking technology also measures a range of supportive and established biomarkers for additional insights and multi-variable analysis.

Small but mighty
- H2O2 contents
- CO2 contents
- Exhaled breath flow
- Exhaled breath temperature
- Exhaled breath relative humidity
Inflammacheck® News
Daily Mail UK: Breathalyser may give doctors a new way to diagnose respiratory conditions in a matter of minutes
The new breathalyser test that spots asthma in minutes and may allow doctors to diagnose other respiratory conditions
Drink, drugs and disease:
The chemistry of breath tests
From roadside tests to diagnosing Covid-19, Clare Sansom looks at how breathalysers have developed.

